What I’ve Learned:
“Radioisotopes: When they have a meltdown, you might, too.”
Chemical elements are exactly like people: there are almost two hundred of them, and only a handful you’d want to invite to a dinner party.
(Okay, it’s possible there are more than two hundred people. But the second part still stands.)
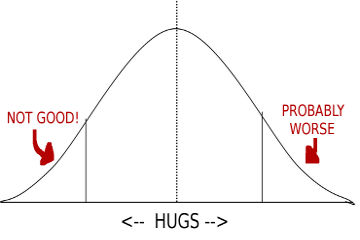
The other way elements are like people is that they both have baggage. With people, it’s a messy divorce, or a predilection for making their pets wear sweaters. Or being an outlier on the Bell curve charting “frequency of parental hugs”.
With elements, it’s neutrons. Nothing else. Just neutrons, little uncharged subatomic bits of schmutz. You would think that would take all the drama out of having baggage.
You would be wrong. It’s like the Real Housewives of the Periodic Table down there. Here’s why:
You can dump extra neutrons onto an atom, no matter how small. Take hydrogen, for example — the runtiest little element of all. It’s got just one proton — the other bit of atomic schmutz that has a positive charge, to offset the negative electron circling the nucleus — and no neutrons at all. Hydrogen is simple that way, like a monk or a wise old hermit or that kid who used to shine shoes on Parks and Recreation, before he got married and buff and went into outer space with that tree and the rodent and the rest of them.
You can pile a neutron onto a hydrogen atom, and it’s mostly fine. This atom is called an isotope, because it’s got more (or less) than the usual number of neutrons — and it’s called deuterium, because that’s what hydrogen atoms with one neutron like to be called.
(I don’t know what sort of nicknames its friends give it. “Deutie” seems fraught with issues. “Deut”, maybe? “Terie”? No idea.)
But deuterium, laden with baggage though it is, is very stable. Makes good decisions. Keeps a steady job. Probably doesn’t even have a therapist — unless it lives in L.A., because pretty much everybody has a therapist there, but still. Deuterium isotopes are chill.
Until you feed them another neutron.
Then those isotopes become tritium, which is a radioisotope. And radioisotopes are atoms where the baggage has gotten to be too much, and it gets unstable. These are the atoms with the crazy eyes, and — like most anyone with too much baggage — they’ll eventually dump it out on those nearby. Explosively.
For radioisotopes, this means radioactive decay — a release of stored energy which brings the atom into a more stable state. Tritium, for instance, decays into an atom of helium-3 (two protons, one neutron), which is completely stable, and fine to invite over for parties or to babysit the kids. But the energy and particles released by decaying radioisotopes can be bad news — or extremely useful, depending on the atom.
Some forms of radioactivity can cause radiation poisoning, cancer or fish with an uncomfortable number of eyes. The rate at which radioisotopes blow their atomic stacks is measured as a half-life — that is, the amount of time it takes for half the atoms in a sample to go completely batshit and decay. Knowing this half-life (and the type of decay — alpha, beta, gamma or other) can come in handy where just the right amount of radioactivity is helpful — like americium-241 used in smoke detectors, or gadolinium-153 used for certain kinds of X-ray tests and osteoporosis screens.
But the most temperamental and energetic radioisotopes — the Kardashians of the atomic world — can cause problems for centuries or longer. Carbon-14 and strontium-90 from nuclear bomb tests, for instance, with a half-life of nearly six thousand years, or nuclear reactor output like cesium-137 and iodine-131 (which can also be used as a cancer treatment, under carefully controlled conditions).
So the next time you decide to dump baggage on someone — or unload some of your own on innocent bystanders — take a moment to think of the radioisotopes. Some of them are just as unstable as you. Only they wig out and break down because of science, and not a tragic hug imbalance. Neat.